TECHNICAL PUBLICATION
New top-lid packaging solution
for rigid blister applications
Download the Polybond brochure (PDF)


CONTEXT
Over the years, multiple materials have been developed to serve as a potential cost effective alternative to Tyvek® based packaging.
The first market developments have been observed in the field of flexible formable packaging where coated and surface treated webs made of a mix of cellulose and synthetic binders or fibers have progressively gained markets shared.
Among manufacturing devices that have been transferred, one can mention drapes, gowns, catheters & IV Sets…
At the end of the year 2000s, a first attempt had been made with the launch of Ovantex® by Oliver, which was a product development associating
- the coating technology of the by-then Oliver company
- to a versatile cellulose-based web material encompassing synthetic fibers & binders to enhance material mechanical strength & cohesion
- the total gauge of the material was of 101 g/m²
- enhanced mechanical properties
- material had been tested and complied with requirements for EO & Irradiation sterilization
In the early 2010s, polymer-reinforced webs were introduced with surface treatment technology helping to drive costs down by removing the need for additional coated layer on base web material.
The solution could be used on regular grades of film, granted that they were comprising a standard layer of Polyethylene for sealing & peeling performances, and has quickly found its market in the field of industrial peel pouch & roll stock packaging solutions for drapes, gowns, lap sponges and IV sets.
Nevertheless, specific requirements of sealing to rigid packaging could not be answered by this new solution, due to:
- seal behavior to PVC, APET, PETG or HIPS being different from the one of a Pe- EVA
- peel angle and mechanical stress when opening a rigid blister being more challenging from aseptic opening requirements than the flexible packaging solutions.
NEW PRODUCT DEVELOPMENT
Hence the development over the last 3 years of new materials aiming at bridging the gap and delivering the right level of performance for blister needs:
- stronger intense cohesion of material
- flexibility and stretch enhancement for large peel angle requirements
- bespoke coated adhesives adapted to the different film natures that can be found on market
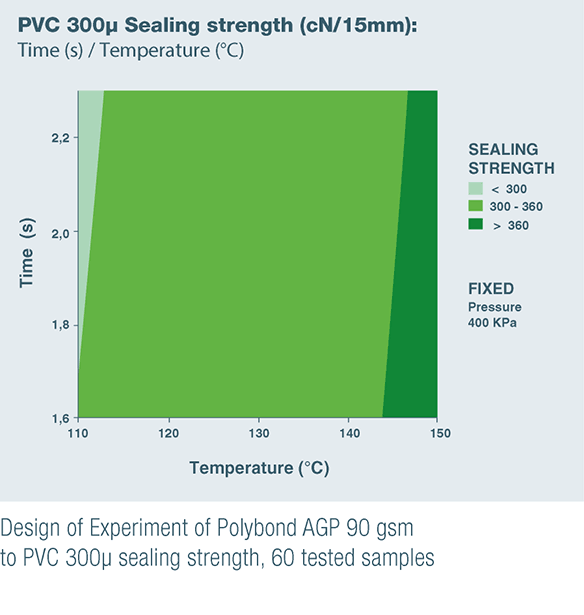
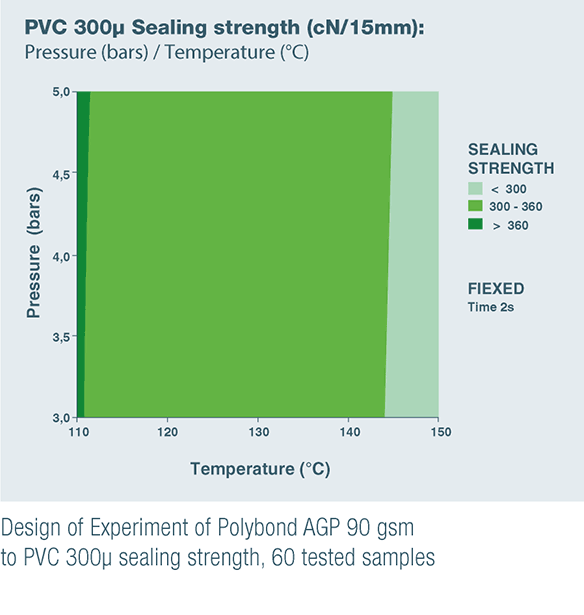
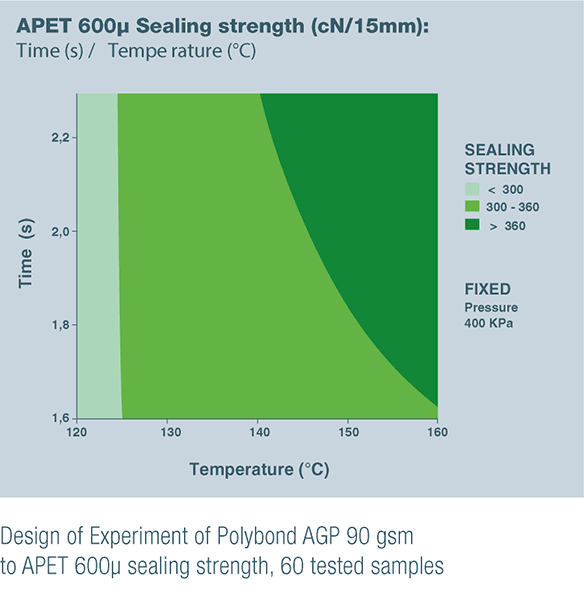
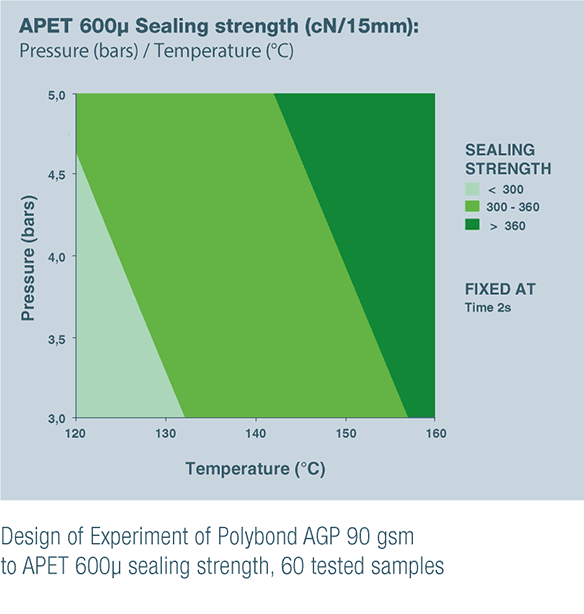
POLYBOND AGP 90 gsm & POLYBOND BHP 85 gsm are the result of these product developments to satisfy unanswered customer needs.
- POLYBOND AGP 90 gsm for sealing to PVC, which is still commonly used in Asia and on some European market, and to APET
- POLYBOND BHP 85 gsm for sealing to HIPS
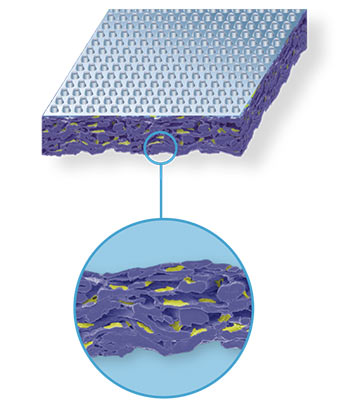
MATERIAL:
fiber mesh of cellulose and polymer binders, with grid coating pattern